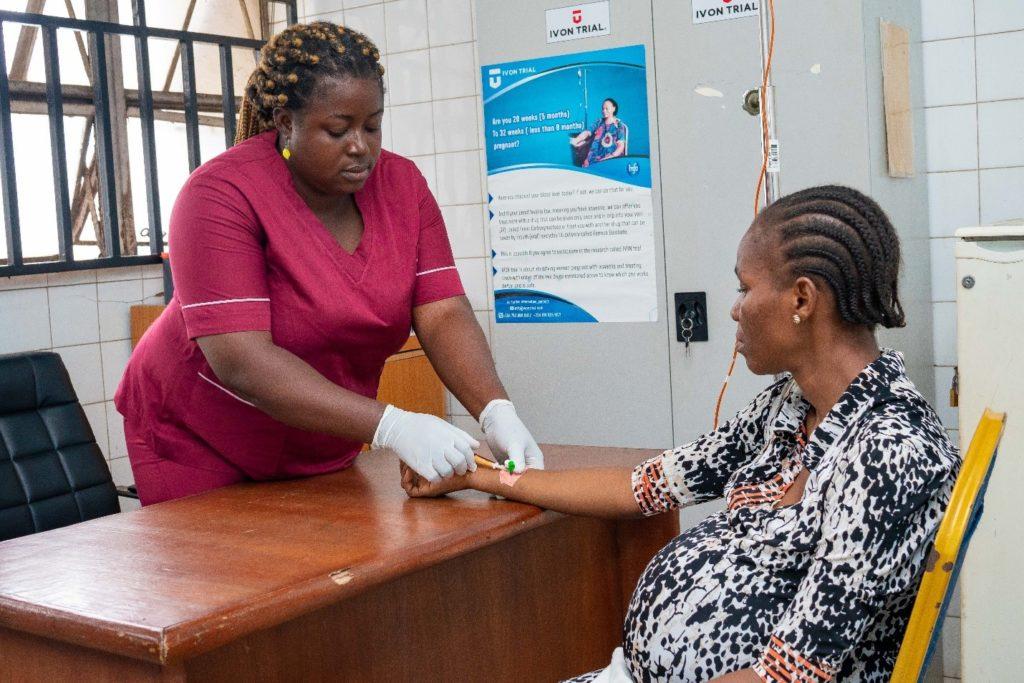
In a bustling primary healthcare center in Lagos, a pregnant woman with severe anemia receives intravenous iron instead of the usual handful of iron tablets. This scene, once unimaginable in Nigeria’s primary care settings, represents a subtle shift in maternal care strategy that could transform how the country addresses one of its most persistent health challenges.
With maternal anemia affecting 56% of pregnant Nigerian women and feeding into the country’s troubling maternal mortality statistics, the IVON-IS project (Intravenous vs Oral Iron for Nigeria – Implementation Science) has been testing whether bringing intravenous iron therapy into the Nigerian healthcare system is both feasible and worthwhile. The project spanned six healthcare facilities across different levels of care—from primary to tertiary—though its success at the primary care level has particularly significant implications for accessibility.
The Iron Dilemma
Anyone who has taken oral iron supplements knows the unpleasant reality: nausea, constipation, and that distinctive metallic taste. For pregnant women, these side effects aren’t merely uncomfortable—they’re deal-breakers. Nigerian studies reveal compliance rates with oral iron dropping as low as 65%, a statistic that isn’t surprising to frontline health workers.
“Women tell us they stop taking the pills because they make them feel worse instead of better,” explains a nurse at Akerele Primary Healthcare Centre. “Then they return with their anemia unchanged or worse, and we start the cycle again.”
Beyond the side effect problem, oral iron demands months of daily pills and repeated facility visits for refills—a tall order in a setting where consistent antenatal care remains a challenge. The clock ticks especially loud for women diagnosed with anemia late in pregnancy, when waiting months for hemoglobin levels to rise isn’t a luxury they have.
The recently published results from the IVON clinical trial, a multi-centre study that enrolled 1056 pregnant women across 11 health facilities in Nigeria, provided compelling evidence that intravenous ferric carboxymaltose (FCM) may offer an alternative. A single 15–20-minute infusion eliminated adherence concerns completely. The trial found that IV iron significantly reduced iron deficiency rates to just 5%, compared to 16% with oral supplements, at 36 weeks’ gestation. For women with confirmed iron deficiency, anemia rates dropped substantially, and no serious drug-related adverse events were observed among 527 pregnant participants who received FCM.
From Trial to Real World
The three-year IVON-IS study complements the IVON trial by translating evidence into real-world practice. While the IVON trial confirmed the clinical benefits of IV iron, IVON-IS assessed the feasibility of implementing it across various healthcare settings in Nigeria.
In February 2025, stakeholders gathered in Lagos to examine the outcomes of this promising intervention when it encountered the complex realities of Nigeria’s health system across all six implementation sites, spanning primary, secondary, and tertiary facilities.
“The clinical benefits were clear,” says Professor Bosede Afolabi, Principal Investigator of the IVON-IS project. “But the question was always: could this intervention work across different levels of our healthcare system, particularly at primary healthcare centers where most women first seek care?”
The results were encouraging across all facility types, with perhaps the most significant impact at the primary care level. At Akerele Primary Healthcare Centre, 78 women successfully received IV iron without severe adverse events—a breakthrough for a facility that previously had to refer similar cases to overcrowded secondary facilities for blood transfusions.
Dr. Sijuade Sanusi, a medical officer at Akerele Primary Healthcare Centre, notes the difference: “Now we can provide effective treatment on-site, which is particularly valuable for women who need rapid improvement in hemoglobin levels.”
The IVON-IS project’s approach of forming local Implementation Management Teams at each of the six facilities proved crucial. Using the Diagnose-Intervene-Verify-Adjust framework, these teams conducted quarterly bottleneck analysis workshops to troubleshoot in real-time, with solutions tailored to each facility’s specific context.
“The bottleneck analysis united us in problem-solving,” shared one team member. “When we hit obstacles—and there were many—we adapted our approach based on local realities.”
The Reality Check
Despite promising results, several challenges emerged that any scale-up effort would need to address.
Time commitment remains a consideration, though less than initially feared. A single IV FCM dose requires approximately one hour at the health facility—15-20 minutes for the infusion and 30 minutes for monitoring afterward. While this represents a single visit rather than multiple appointments for oral supplementation, it’s still an hour away from work, household duties, and childcare responsibilities.
Unlike oral iron, which requires daily doses for months, IV FCM typically needs only one visit for a complete treatment course. The IVON-IS study found no need for repeat infusions when the correct dose was calculated based on the woman’s weight.
Cost presents a more formidable barrier. During the study, IV iron was provided free of charge to participants; however, sustainability demands a different solution. IV iron is considerably more expensive than oral supplements and currently isn’t covered by Nigeria’s National Health Insurance Scheme.
“This is the elephant in the room,” emphasized a health financing expert at the stakeholder workshop. “Without insurance coverage or significant subsidy, most women simply cannot afford IV iron, regardless of its benefits.”
Staff capacity constraints also proved challenging. While the project demonstrated that primary healthcare workers can safely administer IV iron with proper training, a substantial initial investment in training was required, and staff turnover created knowledge gaps that necessitated continuous reinforcement.
The Path Forward
“IV iron isn’t a silver bullet that will eliminate maternal anemia overnight,” notes Professor Afolabi. “But it offers an important alternative for women who need rapid correction of anemia or cannot tolerate oral iron.”
For IV iron to transition from a promising intervention to a sustainable solution, several key elements must be aligned: local production to reduce costs, integration into health insurance schemes and essential medicines list, streamlined protocols for identifying women most likely to benefit, and continued capacity building for healthcare workers.
As stakeholders digest the findings from all six IVON-IS implementation sites, the implications extend beyond the specific intervention to encompass broader questions about how health systems adapt to new evidence. The experience—particularly the success at the primary healthcare level—underscores that simply introducing a clinically effective intervention isn’t enough. Success demands attention to implementation details that determine whether promising innovations can function across different levels of the healthcare system and actually reach the women who need them most.
Addressing maternal anemia with IV iron is not just about proving it works—it’s about making it work for every woman, regardless of where she seeks care.
The IVON-IS project was funded by the Bill & Melinda Gates Foundation

Pregnant woman receiving the IV iron infusion